Potential best-in-class bispecific antibody for Breakthrough immunotherapy
Both PD-1 and VEGF are well established targets for cancer immunotherapies. However, only a small subset of cancer patients respond to current PD-1 therapies, and many patients do not tolerate the adverse events caused by VEGF inhibition.
Current limitations of
PD-1/PD-L1 inhibitors have been established as the standard of care in various cancer types. The landscape of the overall PD-1/PD-L1 is still expanding 10 years after the first approval in this field. However, only a small portion of the cancer patients respond well to the PD-1/PD-L1 therapies. In addition, as the therapies move to earlier lines of treatment, more patients experience relapsed or recurrent diseases.1
Potentially the best-in-class PD-1/VEGF bispecific antibody as the immunotherapy for the future
By targeting PD-1 and VEGF, AI-081 presents an effective therapeutic strategy for multiple tumor types with unique benefits. As a single therapeutic agent with multiple targets, AI-081 showed strong cooperative interactions that lead to enrichment of the bispecific antibody in tumor microenvironment (TME). As a result, the benefit of blocking both targets is enhanced, and the toxicities decrease due to reduction in non-selective angiogenesis inhibition.
AI-081 bears a head-and-tail design, which is more effective to form interactions with both PD-1 and VEGF.
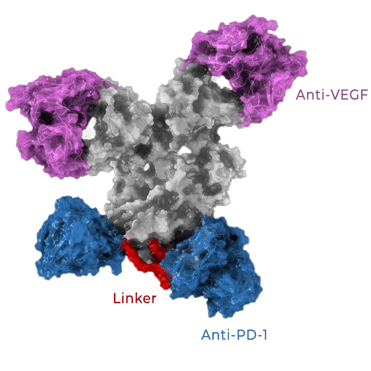
In molecular and cell-based assays, AI-081 consistently shows superior performance over other bispecific antibodies targeting PD-1 and VEGF. In addition, the higher efficacy is also translated into a greater anti-tumor activity in the animal models.
AI-081 is under development in Phase 1 and 2 trials in cancer patients in the US and China.
1. Javed SA, Najmi A, Ahsan W, Zoghebi K. Targeting PD-1/PD-L-1 immune checkpoint inhibition for cancer immunotherapy: success and challenges. Front Immunol. 2024 Apr 10;15:1383456. doi: 10.3389/fimmu.2024.1383456. PMID: 38660299; PMCID: PMC11039846.


